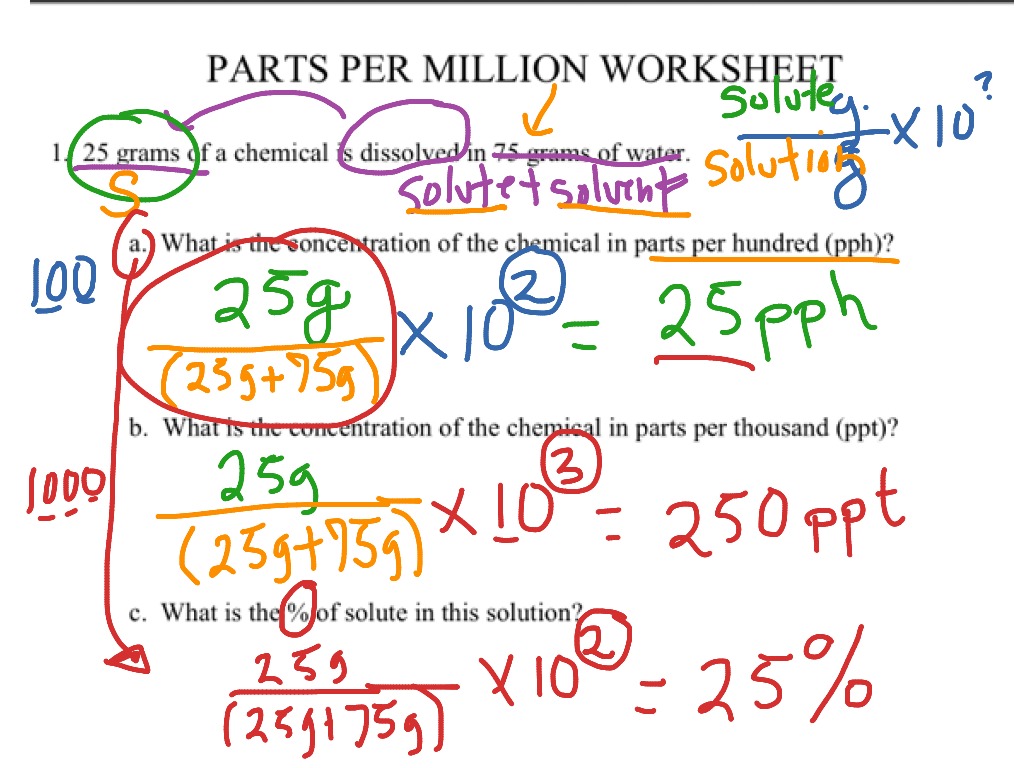
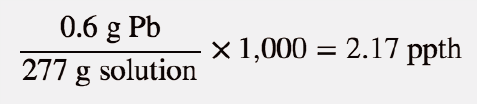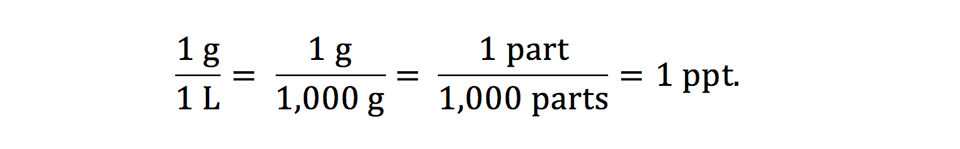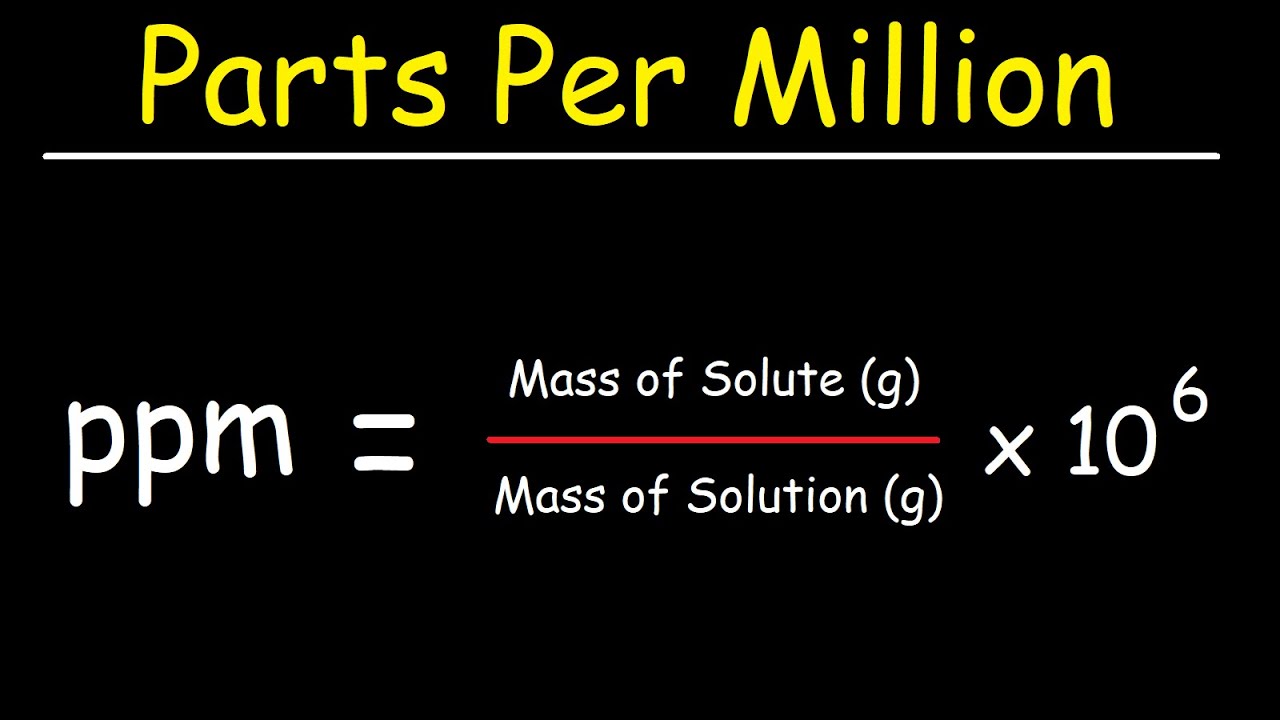SOLVED: Percent by definition is parts per hundred. % (wlw) (mass of solutelmass of solution) x 100. For very dilute solutions, chemists will often use parts per thousand or parts per million (

Twenty Parts Per Thousand — Twenty Thousand Hertz - The stories behind the world's most recognizable and interesting sounds.

Salinity Salinity is the total amount of dissolved material in grams in one kilogram of sea water (Ideal, hard to measure) On average, there is around. - ppt video online download

SOLVED: Percent by definition is parts per hundred: % (wlw) (mass of solutelmass of solution) x 100. For very dilute solutions, chemists will often use parts per thousand or parts per million (